Healthcare Software – Managing Inspection Dates for Medical Devices
On this page you will find information on the following topics:

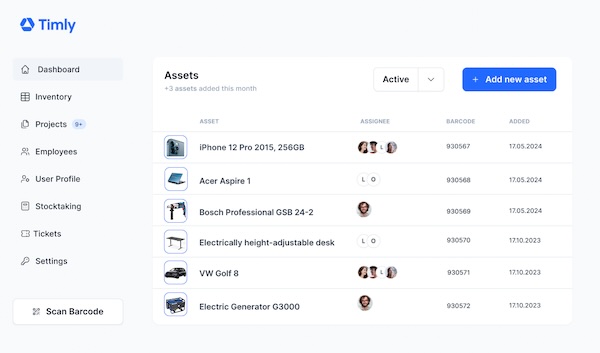
Timly’s inventory software supports you in the management of medical devices
How Healthcare Software Helps You With Medical Device Regulations
Medical device inspections are an important topic for healthcare professionals. There are hardly any areas of work where the correct functioning of devices is so essential. Even small deviations can lead to incorrect treatment and have serious consequences. In addition, workers in healthcare professions are exposed to great stress due to workload and high responsibility.
With healthcare software like Timly, you can ensure compliance with all device regulations. If desired, the software notifies your managers in good time before upcoming inspections. Even the automatic commissioning of maintenance partners via an integrated ticketing system is possible.
In this way, you fulfil your responsibility and relieve nursing staff of tedious administrative tasks. Metrological devices in particular need to be inspected with great care. Their inspection involves determining the measurement accuracy of a device. This is done through calibration or verification. In most cases, the successfully completed process is certified with a test badge and test records. The type, scope and frequency of the tests are specified in special regulations.
What Do Medical Device Inspections Involve?
All medical device inspections must be carried out in accordance with the generally recognised rules of technology for Great Britain, Northern Ireland and the European Union. Details on the recognised rules can be found in the guidelines of the UK government on “Regulating medical devices in the UK“.
The guide includes the legislation that applies in Great Britain as well as the involvement of the MHRA. They also regulate the group of persons who may carry out the tests. This includes competent authorities and institutions as well as employees who fulfil the requirements.
Appropriate certifications are useful. Timly covers this subject area in its entirety. Thus, necessary maintenance and inspection instructions can be stored in the profile of an object. The optional linking of Timly’s personnel management enables those responsible to easily see whether employees intended for an activity have the necessary qualifications.
Asset Management Software in Use by Our Customers
The Timly software is continuously evolving to meet the needs of our customers. In various success stories, we show you how Timly optimizes processes in companies, thereby saving significant effort. With Timly, inventory management becomes child’s play.

Optimized Device Management With Innovative Self-Inventory
SodaStream is the world market leader for water sparkling systems for domestic use and has a lot of IT equipment at its various locations. Many colleagues now work from their home offices. A digital solution for the efficient management of IT end devices became necessary...

Panasonic x Timly: Driving Technological Innovation
One of the most remarkable aspects of human ingenuity is our ability to innovate. Innovation is embedded in the DNA of consumer electronics giant Panasonic, which has diversified into a number of sectors, from heavy industry to construction...

Manage Video Equipment Efficiently Without Much Effort
The Hamburg media company always does outstanding journalistic work and is characterized by independent reporting. In order to maintain journalistic quality, the teams work with highly specialized devices – these need to be managed efficiently...

Smart City Asset Management – Timly in Use at DIGOOH
The core business of DIGOOH Media GmbH in Cologne is to manage digital city light posters (DCLP) for outdoor use in various cities in Germany. The challenge here lies in making the client’s communication message always available at the right time, in the right place...
(No credit card required)
Regulating Medical Devices in the UK
The Medicines and Healthcare products Regulatory Agency (MHRA) specifies the ordinance with regard to the inspection intervals to be observed. These are specific to each device and range from one to six years.
However, there are also exceptions for devices that must be calibrated more frequently under certain conditions. This brief outline already makes it clear that a maintenance planner considerably simplifies deadline management.
How Often Do Devices Need to Be Inspected?
Timly can answer the question of when the next check is due and whether the previous checks have been positive with a simple scan of a QR code on the device. The software is designed in such a way that it can record any inventory. This is ensured by dynamically adaptable profiles.
A sticker with a QR code serves as an identification feature. The data is stored in a cloud provided by the provider, whose secure administration – in compliance with the GDPR standards – is already included in the running costs. With an internet-enabled device, for example a smartphone, the nursing staff can easily access the profile of each device via the app optimised for mobile devices.
Over 600 Companies, Schools and Cities Rely on Timly
What Devices Need To Be Inspected?
Since medical devices are used on the human body, they need to be treated with special care. Hence, they must be inspected on a regular basis. Blood glucose meters, for instance, are a special case because they are given to patients. This gives them the opportunity to measure their blood glucose level at home. The control is carried out by a test measurement using a control fluid.
In care facilities or when handing them out to patients for their own use, a healthcare software with a maintenance planner ensures that those affected and those responsible are notified in good time – for example, by e-mail.

The healthcare software by Timly notifies you about upcoming inspections
Other measuring devices, such as the blood pressure monitor, are part of the standard equipment of many outpatient and inpatient care facilities. The blood pressure monitor is probably one of the diagnostic devices most frequently used in the everyday work of caregivers. Due to constant use and often changing locations, they are subject to a certain amount of wear and tear. With Timly, you can make personal assignments. Defects can be reported in the field via app.
Similar Blog Posts:
Book an online demo - free and without obligation - or create your free trial account directly.