Digital Inventory Management for Healthcare Facilities

Inventory Everything
- Digitally capture every type of inventory.
- Flexible category & attribute structure.
Assign Everything
- Assign items to resources via a barcode scan.
- Historically traceable and audit-proof.
Automate Maintenance
- Manage defects & issues via a ticketing system.
- Monitor performance & consumption data.
Expected Results
- Central overview of your entire inventory.
- Ensure operational readiness of your inventory.
Record and Manage All Inventory Centrally
Record and manage all devices and equipment centrally, including accessories. From EKG and ultrasound machines to laboratory equipment, beds, furniture, and medical consumables. With a digital item file, all relevant information for each item – such as documents, manuals, maintenance history, locations, and responsible personnel – is stored in one place.

All Items

Consumables

All Locations
Keep track of your entire company inventory, no matter how many locations it moves between.

Import Existing Data
Assign Inventory to Any Type of Resource
- Who is currently using Infusion Pump X?
- When does the medical device in OR 2 need to be inspected?
- Which treatment room is the mobile ultrasound device currently in?
- Is there enough material available for next week’s OR schedule?
Digital Maintenance Management for Your Inventory
TICKETING SYSTEM
Centralize All Your Service and Maintenance Requests
Centralize all your service, support, and maintenance requests. The ticketing system automatically creates, prioritizes, and schedules tasks.
Faster processing of service and maintenance cases
Clear accountability through automatic assignment
Complete documentation of every activity


ITEM PERFORMANCE
Leverage Performance and Consumption Data Intelligently
Connected IoT sensors provide real-time data, detect issues early, and help plan maintenance optimally.
Real-time overview of your entire inventory
Early warnings for anomalies or defects
More effective planning and utilization of your resources
DIRECT-ACCESS LABELS
Make Item Data Instantly Accessible via QR Codes
No login required – service partners and customers can report damage or issues directly on the item, keeping information up to date and processes smooth.
Instant access to item data via QR code
Always up-to-date information directly on the item
Faster, more transparent service processes

What Our Customers Achieve with Timly
Clinics, hospitals, and laboratories gain significantly greater visibility over their inventory, reduce losses and downtime, and speed up their daily operations. From maintenance and service to the provision of consumables and reporting, you have full control over everything. See for yourself.
Central Overview
- What do we have?
- Where is the inventory?
- Who has it right now?
- Who needs it where?
Operational Readiness
- When is the next service due?
- Which service partner?
- Service history?
- Documentation?
Time Savings
- Access from anywhere
- Transparent processes
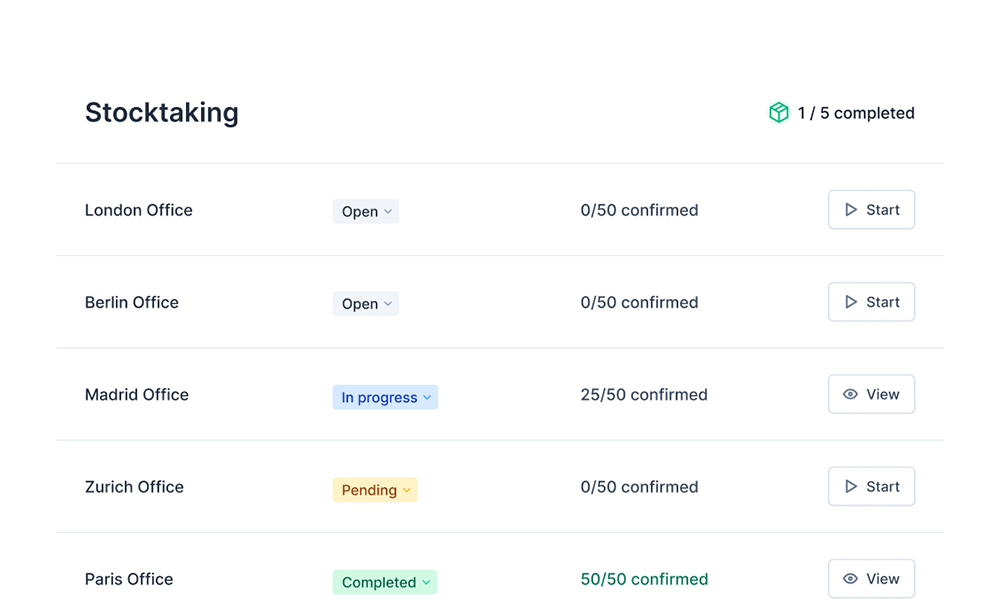
- Digital stocktaking
- Reports
Resource Efficiency
- Fewer outages
- Lower search costs
- Increased employee accountability
- Predictable ROI
ROI of Timly Software for Your Company

Get Started Easily, Simplify Processes
"Timly is very easy to use and simplifies many of our processes. The support from the Timly team is efficient and solution-oriented. In short: Get started now!"
Giulia Agthe | Project Manager

Flexible, Reliable, Customer-Oriented
"Timly has proven to be a very reliable and flexible partner in all areas. Requests and needs are addressed and implemented quickly and efficiently."
Thomas Weiß | Home Director

Intuitive Inventory Management for All Laboratories
"Timly is a great tool for easily inventorying a wide variety of items. It is intuitive to use and offers many options for inventory management. Now everyone has a quick overview of their devices across all labs, can manage reactor maintenance, and we have a fully digital solution."
Jan Wendel | Chemical Technician

Digital Pioneer in Beauty & Dermatology
"We are leaders in innovation in the beauty and dermatology sector, growing rapidly and recognized as pioneers in many areas. We also want to live up to this role digitally, which is why we rely on the cloud-based quality management solution from Timly Software AG."
Bruno Schulze | Owner
Features of the Timly Inventory Solution
Ticketing System
Digital Stocktaking
Live Tracking
Barcode Scanner
Versatile Interfaces That Connect Your Systems
Frequently Asked Questions About Inventory Management
Get answers to the most important questions about Timly – the digital inventory management solution for your healthcare facility. Here you’ll find helpful information to make getting started and using the system easier.
Timly consolidates all medical devices and equipment into a central digital inventory overview, with each item assigned to its area, room, and location. Using QR codes, the mobile app, and flexible filters, authorized staff can always see where each device is, its condition, and which department is responsible, even across multiple locations.
Yes. With Timly, you can centrally schedule and manage maintenance and other legally required inspections using the integrated maintenance and inspection planner. For each medical device, you can set recurring inspection intervals, assign responsible staff, receive automatic reminders, and securely document all inspection reports and records in the digital item file.
Nurses and doctors simply scan the device’s QR code or search for it in the Timly app to instantly see its current location, assigned user, and status. Thanks to mobile access, GPS tracking, and assignment to individuals, rooms, or wards, it becomes clear within seconds where a device is and who is using it.
The software is hosted in secure data centers that meet international standards, including ISO 27001, ISO 27017, and ISO 27018 for cloud security and data protection. Combined with encrypted data transmission, strict access controls, and a data processing agreement in line with U.S. privacy regulations, Timly provides a high level of data protection.
This makes the solution explicitly suitable for healthcare environments and safe for handling sensitive patient information, without the need to expose your practice or clinic network externally.
Timly offers a finely granular, role-based permission system, allowing you to create custom roles for medical technology, nursing, administration, and IT with precisely tailored access rights.
You can define which groups can access which modules, whether they can view or edit data, and even restrict access to specific locations, areas, or device categories, ensuring that sensitive information is only visible to the appropriate personnel.
Yes. Timly is already used by numerous healthcare facilities, including hospitals, medical practices, emergency services, and organizations such as the German Red Cross, SOS Doctors Zurich, and Bayer. In case studies, these clients report reduced losses, better transparency of medical equipment, and significantly more efficient processes in maintenance, logistics, and inventory management.
Yes. You can either book a free online demo with an expert or start a non-binding 10-day trial of the Timly inventory software yourself and test the features with your own data, completely risk-free.
End Inventory Chaos on the Ward – Manage Your Equipment and Supplies Digitally and Transparently Now!